



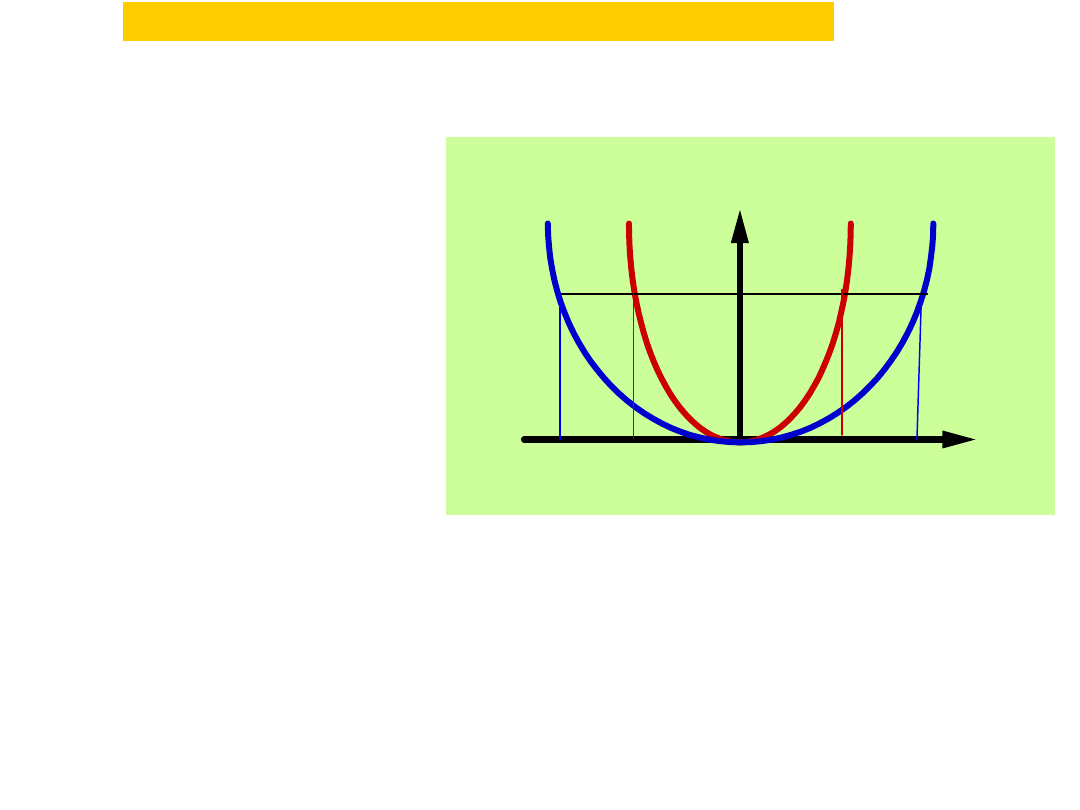
V(x) = 1/2k2x2
V
x
-A2
A2
k1>k2
E
V(x) = 1/2k1x2
-A1
A1
k
1
k
2
Appendix
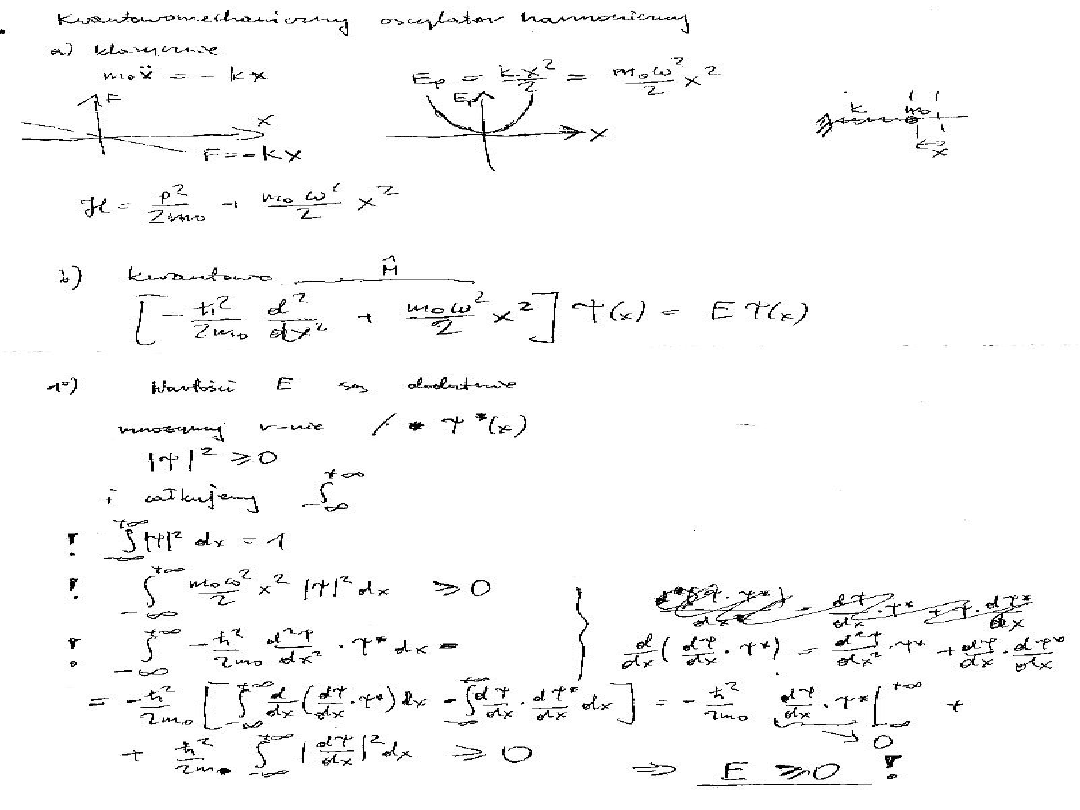
: Classical harmonic oscillator
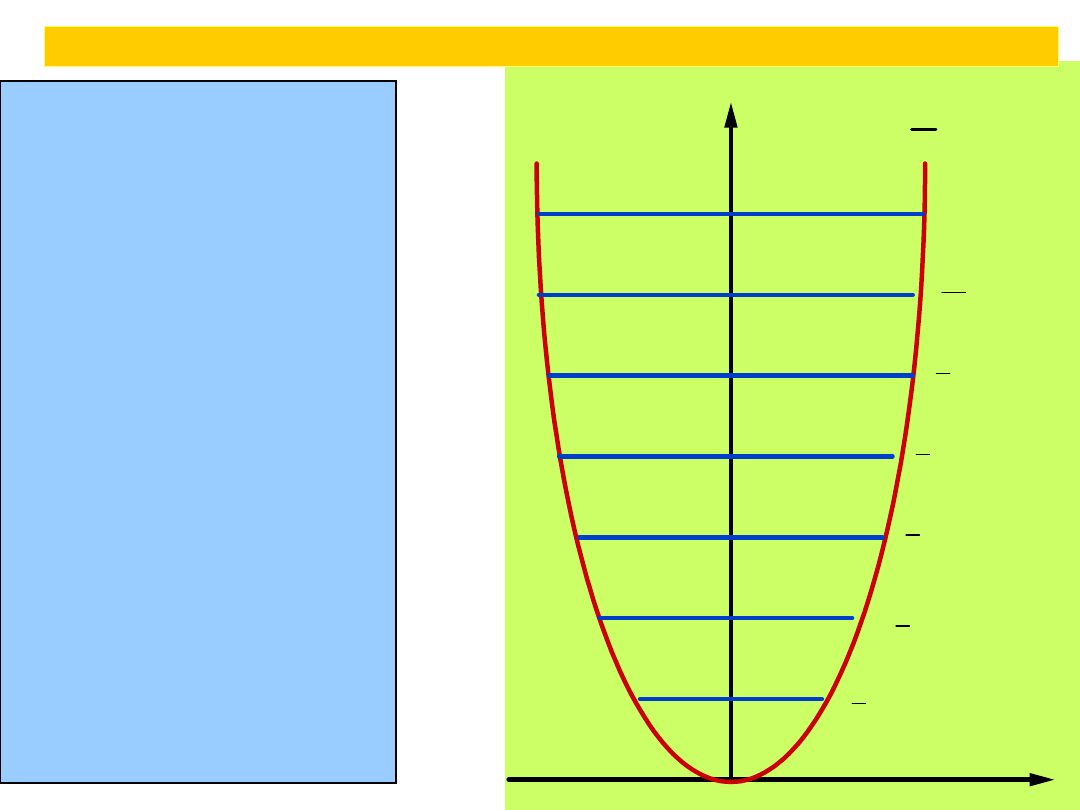
The parabolic potential energy V =
1
/
2
kx
2
a harmonic oscillator, where x is
the displacement from equilibrium. The
narrowness of the curve depends on
the force constant k: the larger the
value of k, the narrower the well.
E
1
2
h
3
2
h
5
2
h
7
2
h
x
9
2
h
11
2
h
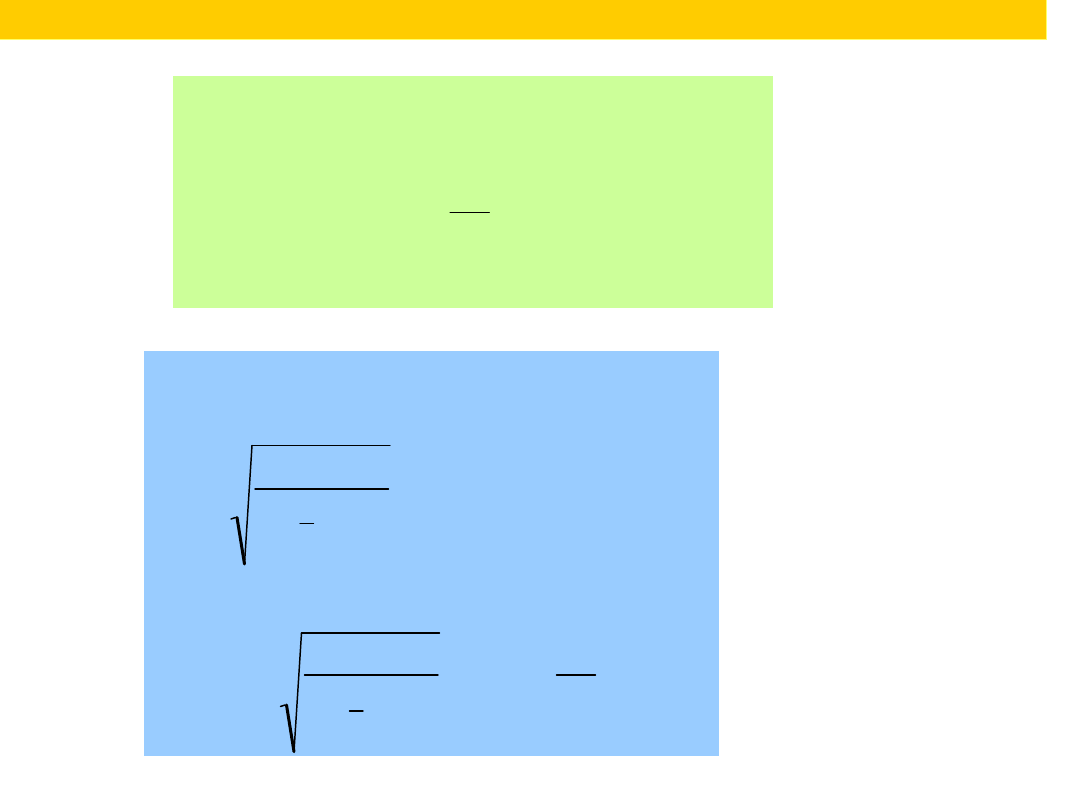
The energy
levels of a
harmo
nic
oscillator
are evenly
spaced with
separation
·, with =
(k/m)
1/2
.
Even in its
lowest state,
an oscillator
has an
energy
greater
than zero.
Harmonic oscillator...Quantum mechanically .Energy levels
v 0
v 1
v 2
v 3
v 4
v 5
v 6
E h
(
1
2
v)
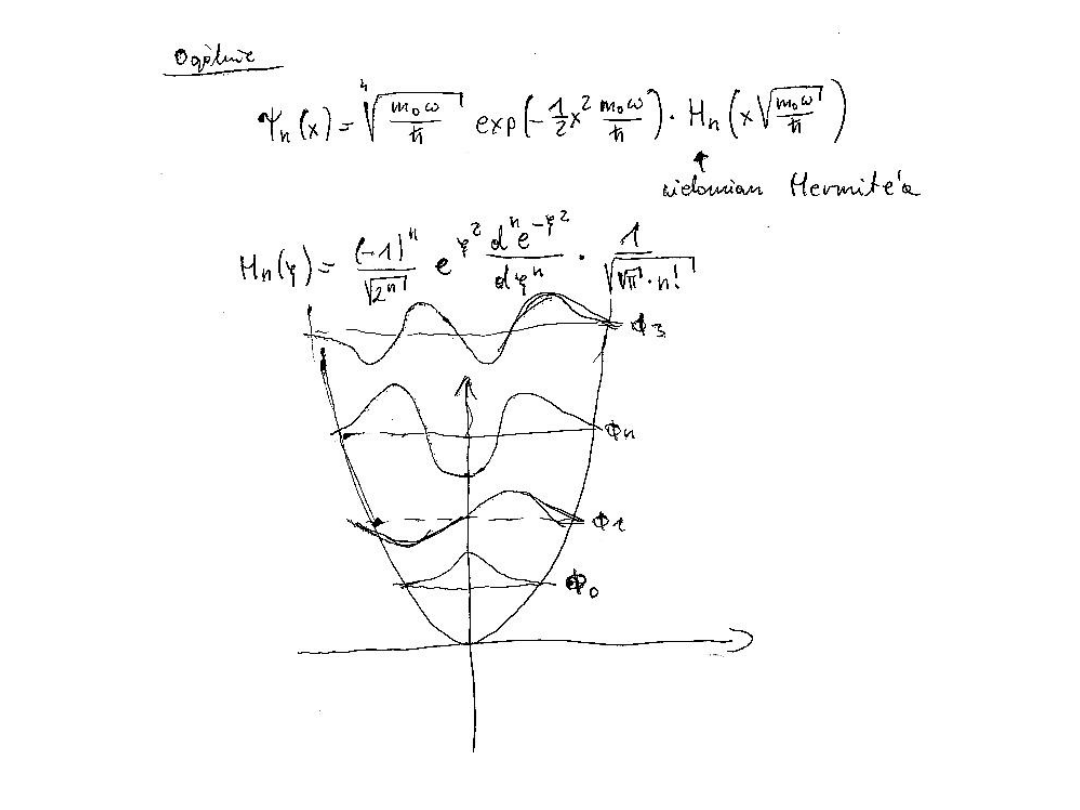
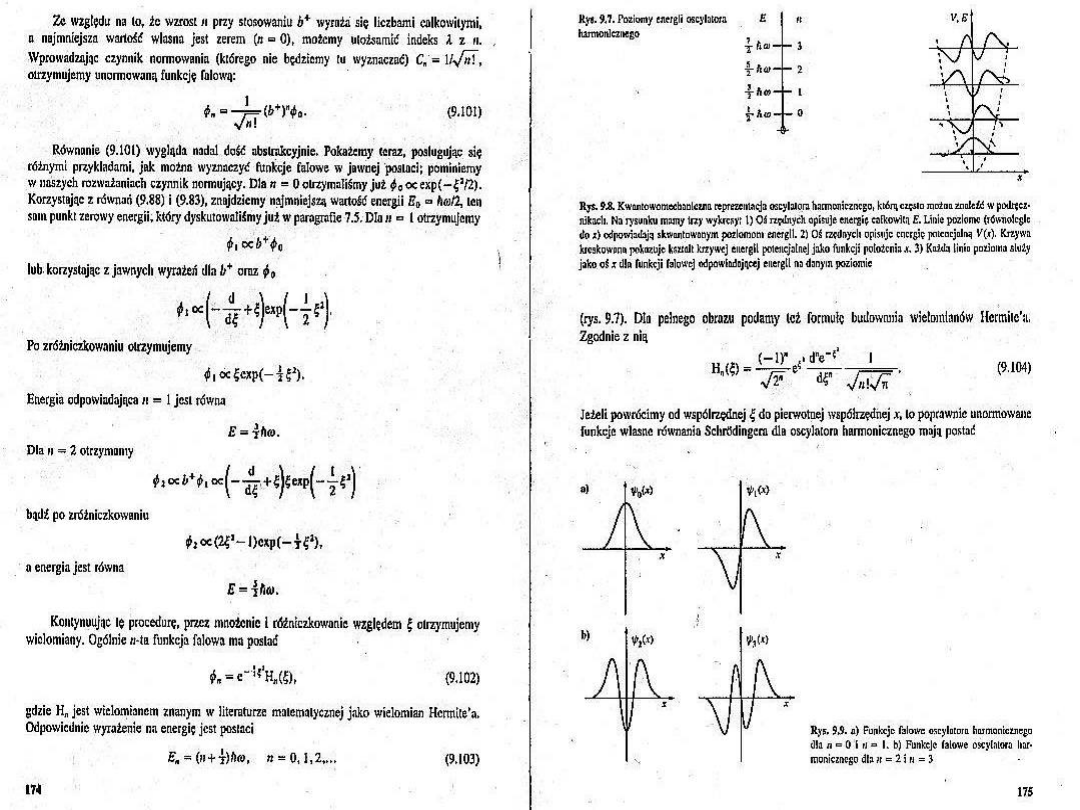
We have the general solution
v
(x)N
v
exp
y
2
2
H
v
(y) ; y=x/
Harmonic oscillator...Quantum mechanically.... Wavefunction
It is readilly shown that
N
v
1
1
2
2
v
v!
so
v
(x)
1
1
2
2
v
v!
exp
y
2
2
H
v
(y)
Harmonic oscillator...Quantum mechanically.... Wavefunction
_________________________
v H
v
_________________________
0 1
1 2y
2 4y
2
-2
3 8y
3
- 12y
4 16y
4
- 48y
2
+12
5 32y
5
-160y
3
+120y
6 64y
6
- 48y
4
+72y
2
-120
_____________________________
Hermit
polynominals
Note
that H
v
for v odd (1,
3,5,7,..)
is odd
: H
v
(y) = - H
v
( y)
Note
that H
v
for v even (0,
2,4,6,8...)
is even
: H
v
(y) = H
v
( y)
Harmonic oscillator...Quantum mechanically.... Wavefunction
v
(x) N
v
exp
y
2
2
H
v
(y)
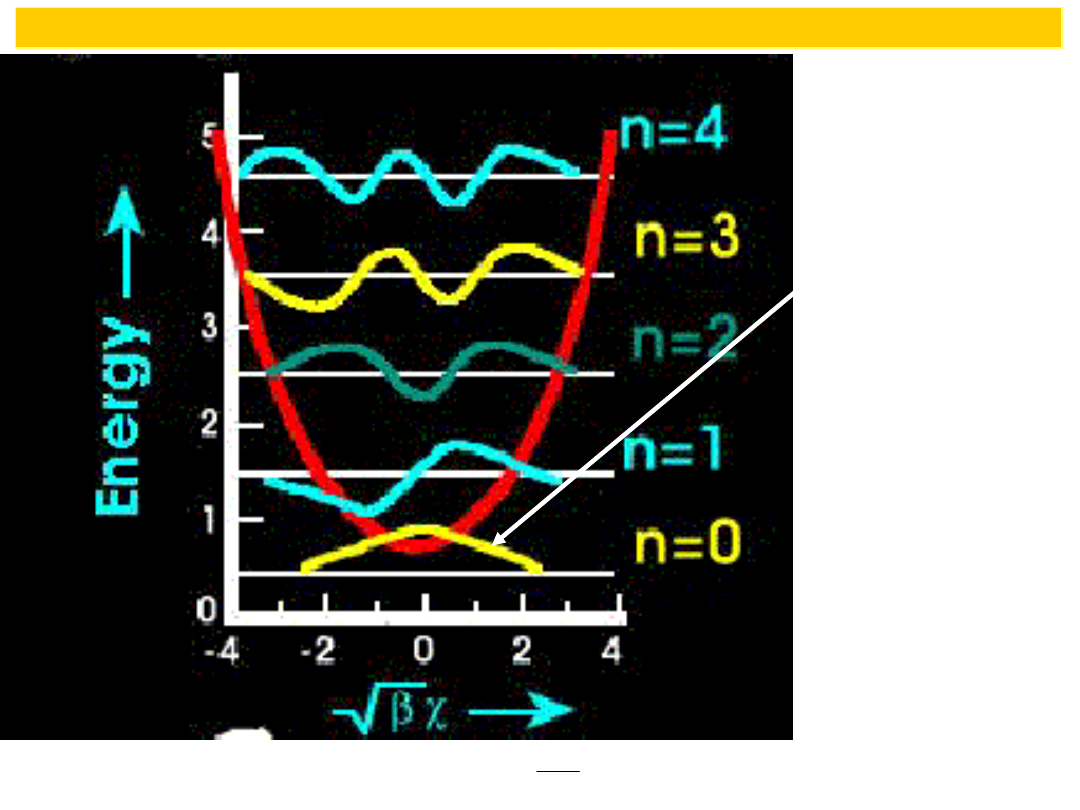
Particle can
be found
outside
clasical region
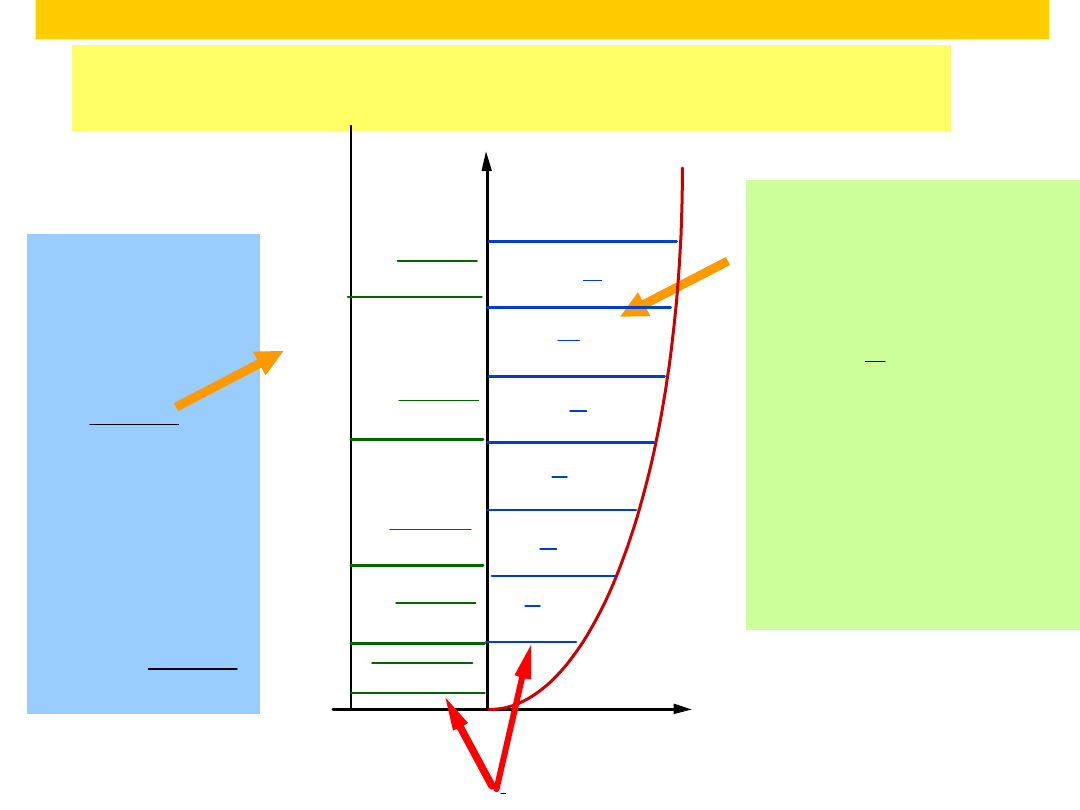
Comparison
of energy levels in harmonic oscillator
and particle in a box
Energy levels for
harmonic oscillator
E =h(
1
2
v)
v 0,1,2,3
Spacing
E h
Energy levels
in particle in
box
E =
n
2
h
2
8mL
2
n 1,2,3
E
(2n 1)
h
2
8mL
2
E
1
2 h
3
2
h
5
2 h
7
2 h
9
2 h
11
2 h
v=0
v=1
v=2
v=3
v=4
v=5
Harmonic oscillator
Particle-in-box
n=1
n=2
n=3
n=4
n=5
h
2
8mL
2
4 h
2
8mL
2
9 h
2
8mL
2
16 h
2
8mL
2
25 h
2
8mL
2
Zero-point Energy
Harmonic oscillator...Quantum mechanically .Energy levels
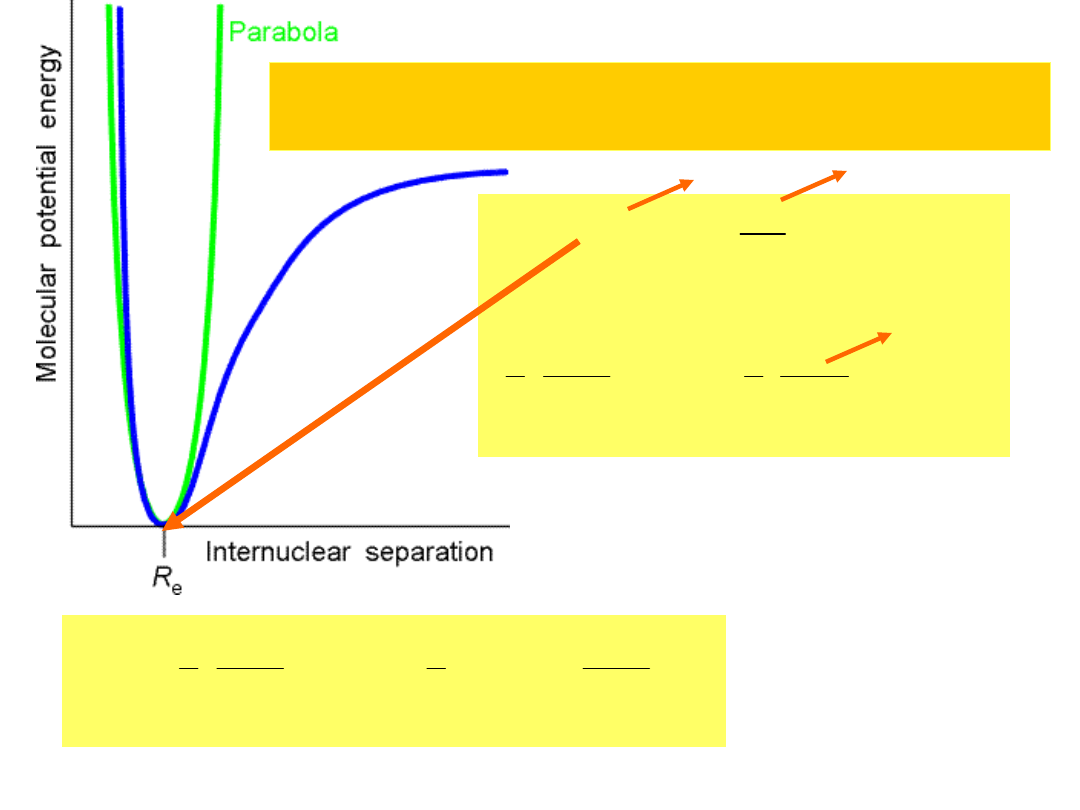
Harmonic oscillator...Quantum mechanically..
Vibration Spectroscopy
V(R)V(R
e
) (
dV
dR
) R
e
1
2
(
d
2
V
dR
2
)R
e
2
1
8
(
d
3
V
dR
3
)R
e
3
...
Taylor expansion
0
small
0
V(R)
1
2
(
d
2
V
dR
2
)R
e
2
1
2
kR
e
2
;(
d
2
V
dR
2
)k
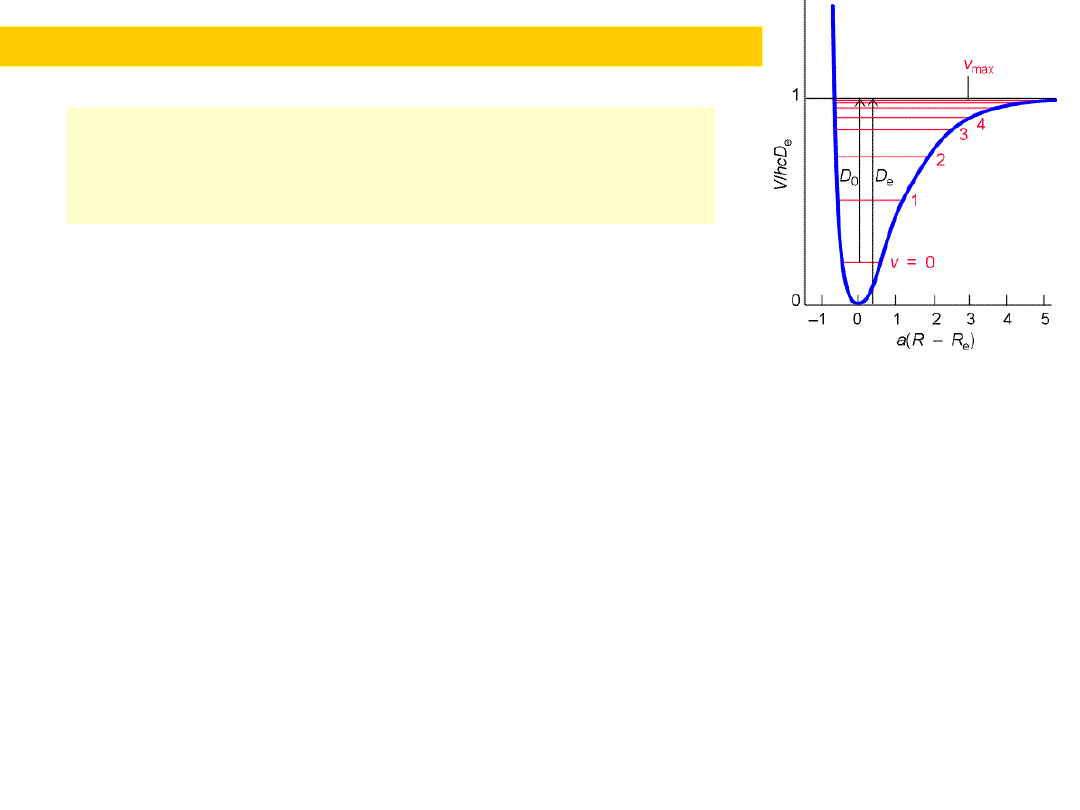
Harmonic oscillator...Quantum mechanically
We note relation between bond energy D ;
bond order and force constant k
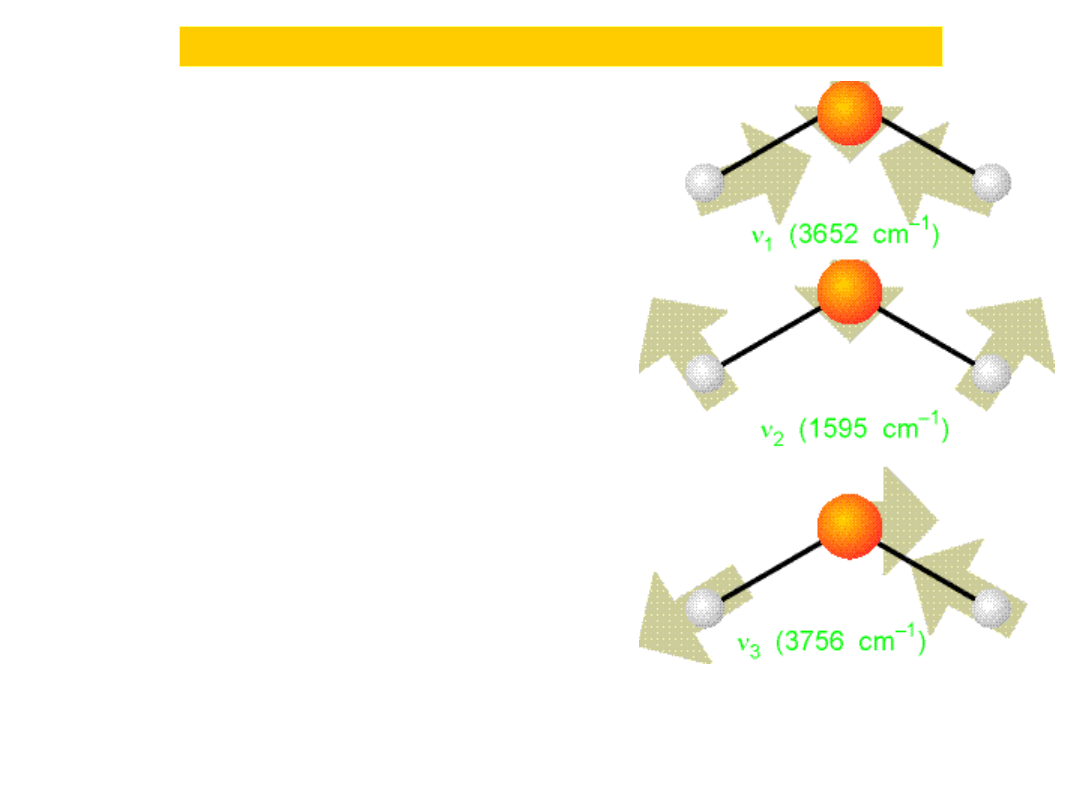
Harmonic oscillator...Quantum mechanically
The three
normal
modes of
H
2
O. The
mode v
2
is
predominant
ly bend
ing,
and o
ccurs
at lower
wavenumber
than the
other two.
Document Outline
- Slide 1
- Slide 2
- Slide 3
- Slide 4
- Slide 5
- Slide 6
- Slide 7
- Slide 8
- Slide 9
- Slide 10
- Slide 11
- Slide 12
- Slide 13
- Slide 14
- Slide 15
- Slide 16
Wyszukiwarka
Podobne podstrony:
EŚT 07 Użytkowanie środków transportu
07 Windows
07 MOTYWACJAid 6731 ppt
Planowanie strategiczne i operac Konferencja AWF 18 X 07
Wyklad 2 TM 07 03 09
ankieta 07 08
Szkol Okres Pracodawcy 07 Koszty wypadków
Wyk 07 Osprz t Koparki
zarządzanie projektem pkt 07
Prezentacja NFIN 07
6 Zagrozenia biosfery 07 05 05
więcej podobnych podstron